Calculating formal charge in atoms7/31/2023 
Example: CO 2 is a neutral molecule with 16 total valence electrons. To calculate the formal charge on nitrogen there are three pieces of information that you need to know: the group number (number of valence electrons), the.Note: The total formal charge in a molecule should be as close to zero as possible, with as few charges on the molecule as possible After adding up all the formal charges throughout the molecule the result is a total formal charge of +1, consistent with the charge of the molecule given in the first place. Thus, using the definition of formal charge, hydrogen has a formal charge of zero (1-(0 + ½ × 2)) and nitrogen has a formal charge of +1 (5−(0 + ½ × 8)). There are no lone pairs of electrons left. 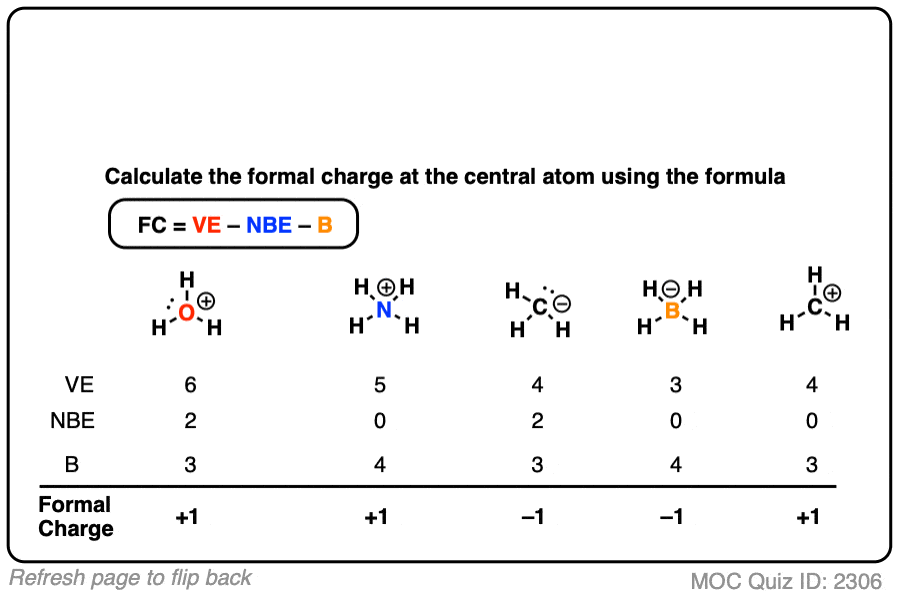
Drawing the Lewis structure gives an sp 3 (4 bonds) hybridized nitrogen atom surrounded by hydrogen. The final total is 8 total electrons (1 × 4 + 5 − 1). By using the vertical groups of the atoms on the periodic table it is possible to determine that each hydrogen contributes 1 electron, the nitrogen contributes 5 electrons, and the charge of +1 means that 1 electron is absent. Other related concepts are valence which counts number of electrons that an atom uses in bonding and coordination number, the number of atoms bonded to the atom of interest.Īmmonium NH 4 + is a cationic species. single bonded oxygen in NO 2 - FC = 6 - 6 - 2/2 = -1Īn alternative method for assigning charge to an atom taking into account electronegativity is by oxidation number.double bonded oxygen in NO 2 -: FC = 6 - 4 - 4/2 = 0.Nitrogen in the nitro group NO 2 -: FC = 5 - 2 - 6/2 = 0.carbon in methane: FC = 4 - 0 - 8/2 = 0.When determining the correct Lewis structure (or predominant resonance structure) for a molecule, the structure is chosen such that the formal charge on each of the atoms is minimized. Better weighing performance in 6 easy steps
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
